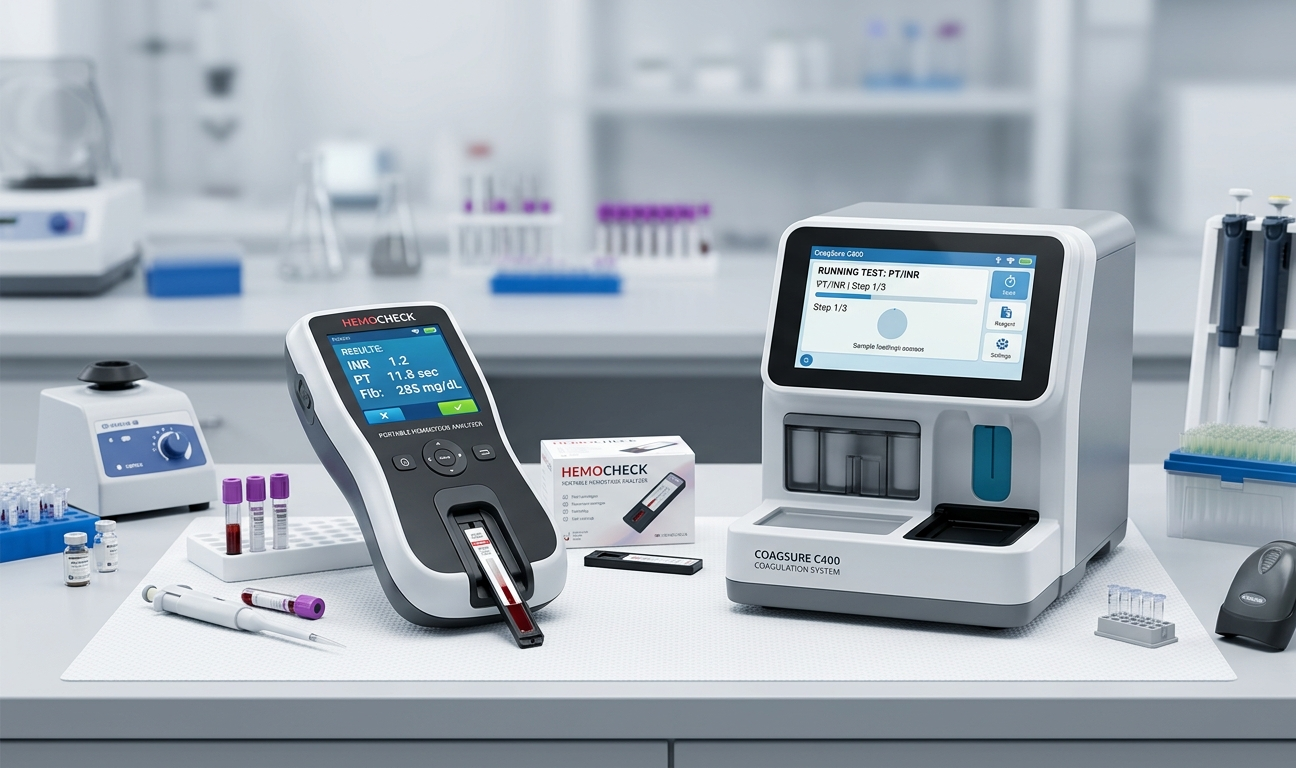
The Portable Hemostasis Analyzers Market is in a period of rapid evolution, with continuous product launches, technological breakthroughs, and strategic corporate activities reshaping the competitive landscape. Innovation remains the cornerstone of growth in this sector, as manufacturers race to develop devices that offer greater accuracy, broader test menus, enhanced connectivity, and improved user experience. The market is characterized by a vibrant ecosystem of established medical device companies, innovative startups, and academic research institutions collaborating to advance the science and technology of portable coagulation testing. These collective efforts are driving a new generation of hemostasis analyzers that are set to redefine point-of-care diagnostics.
An analysis of Portable Hemostasis Analyzers Market recent developments reveals a wave of transformative activities across product development, regulatory approvals, strategic partnerships, and market expansions. Leading manufacturers have announced next-generation portable analyzers featuring artificial intelligence-powered result interpretation, expanded coagulation test panels, and seamless integration with digital health platforms. Regulatory agencies in North America, Europe, and Asia-Pacific have granted approvals for several innovative products, accelerating market availability. Strategic mergers and acquisitions have further reshaped the competitive landscape, as large diagnostics companies seek to strengthen their portfolios through the acquisition of specialized coagulation testing companies. These developments collectively signal a period of dynamic transformation and robust growth.
Ask for Sample Report @ https://www.theinsightpartners.com/sample/TIPRE00025420
New Product Launches and Next-Generation Analyzers
The past several years have seen a significant surge in new product launches in the portable hemostasis analyzers market, reflecting intense R&D investment by manufacturers. Next-generation devices are characterized by their ability to perform comprehensive coagulation panels from small blood samples with minimal operator intervention. Innovations in reagent cartridge technology have simplified testing protocols, reducing the risk of user error and improving result reproducibility. Several manufacturers have launched devices with expanded connectivity features, including real-time Bluetooth data transmission to electronic health records and dedicated clinical management software platforms, facilitating seamless integration into hospital workflows.
Artificial Intelligence and Machine Learning Integration
The integration of artificial intelligence and machine learning algorithms into portable hemostasis analyzers represents one of the most significant recent technological developments in the market. AI-powered interpretation algorithms can analyze complex coagulation profiles, identify patterns indicative of specific hemostatic disorders, and generate treatment recommendations based on established clinical protocols. These intelligent decision support capabilities are particularly valuable in emergency and critical care settings, where clinicians may need to make rapid decisions under significant time pressure. Early clinical studies evaluating AI-assisted hemostasis interpretation have demonstrated promising results, with AI algorithms showing performance comparable to experienced hematologists in identifying coagulation abnormalities.
Strategic Acquisitions and Corporate Partnerships
Strategic acquisitions and corporate partnerships have been a defining feature of the portable hemostasis analyzers market in recent years. Major diagnostics companies have pursued targeted acquisitions to gain access to novel coagulation testing technologies, intellectual property, and established market positions. These acquisitions are typically aimed at expanding the acquirer's point-of-care diagnostics portfolio and accelerating entry into high-growth market segments. Strategic partnerships between medical device manufacturers and digital health companies are also increasingly common, driven by the shared objective of creating integrated coagulation management ecosystems that combine portable testing hardware with advanced data analytics and telemedicine capabilities.
Regulatory Milestones and Market Authorizations
Recent years have seen important regulatory milestones in the portable hemostasis analyzers market, with several novel devices receiving market authorization in major geographies. In the United States, the FDA's De Novo pathway and 510(k) clearance process have enabled innovative point-of-care coagulation testing platforms to reach the market with relatively streamlined regulatory review. In Europe, the transition to the In Vitro Diagnostic Regulation (IVDR) has introduced more stringent requirements for clinical evidence, prompting manufacturers to invest in robust clinical validation programs. These regulatory developments are ultimately raising the quality and performance standards of portable hemostasis analyzers available to healthcare providers.
Clinical Research and Evidence Generation Activities
An increasing body of clinical research is supporting the expanded use of portable hemostasis analyzers across diverse clinical applications. Multicenter randomized controlled trials evaluating viscoelastic testing-guided transfusion protocols have demonstrated significant reductions in blood product usage and improved patient outcomes in cardiac surgery, trauma, and liver transplantation. Real-world evidence studies are providing complementary insights into the clinical and economic value of portable coagulation testing in routine clinical practice. These evidence generation activities are strengthening the case for point-of-care hemostasis testing and supporting the incorporation of portable analyzers into clinical practice guidelines and institutional protocols.
Related Report:
1) Eye Glasses Market Size, Trends & Growth by 2034
2) 3D Printing Medical Devices Market Share, Size & Demand by 2034
3) Insulin Pumps Market Size, Growth & Demand by 2034
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Information –
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean German Japanese French Chinese Italian Spanish