The United States continues to play a central role in advancing pharmaceutical preservation technologies, supported by a strong biotechnology ecosystem and increasing production of complex therapeutics. Freeze drying has become a critical process for stabilizing temperature sensitive drugs, particularly biologics and vaccines, which require long shelf life and consistent performance. The growing reliance on specialized service providers reflects the need for efficiency, compliance, and scalability across pharmaceutical manufacturing.
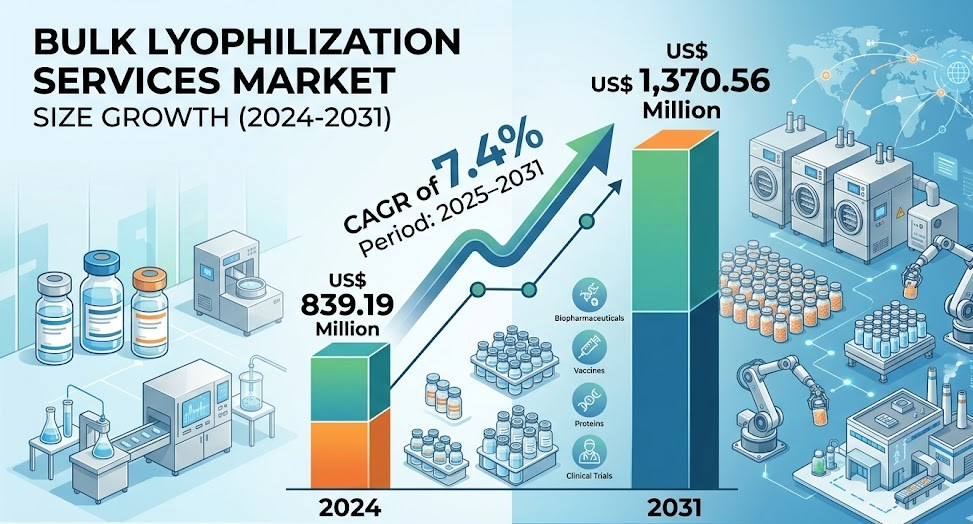
The US Bulk Lyophilization Services Market is evolving steadily as demand for biologics and advanced therapies rises across the country. While the global bulk lyophilization services market is projected to grow from US$ 839.19 million in 2024 to US$ 1,370.56 million by 2031 at a CAGR of 7.4%, the United States represents a significant share of this expansion. The country is also expected to register a CAGR of 7.8% during the forecast period, driven by robust outsourcing trends and continuous innovation in freeze drying technologies.
Expanding Biologics Pipeline Supporting Demand
The increasing development of biologics such as monoclonal antibodies, vaccines, and gene therapies is a primary driver for lyophilization services in the United States. These therapies are highly sensitive in liquid form and require advanced preservation to maintain their structural integrity and efficacy. Bulk lyophilization converts these formulations into stable powders, reducing dependency on cold chain logistics and enabling wider distribution.
The United States leads in clinical research and drug approvals, which directly contributes to higher demand for freeze drying services. Pharmaceutical companies are focusing on improving product stability and extending shelf life, particularly for injectable drugs and vaccines. This trend is expected to sustain long term demand for bulk lyophilization solutions.
Download Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00041488
Rising Preference for Outsourcing to CDMOs
Outsourcing continues to shape the US pharmaceutical manufacturing landscape. Companies are increasingly partnering with contract development and manufacturing organizations to handle complex processes such as bulk lyophilization. This approach helps reduce capital investment, operational complexity, and regulatory burden associated with in house facilities.
CDMOs provide flexible and scalable solutions, enabling pharmaceutical companies to accelerate product development and commercialization. The ability to access specialized expertise and advanced infrastructure makes outsourcing an attractive option. As a result, collaborations between drug developers and service providers are becoming more frequent, strengthening the overall service ecosystem in the United States.
Technological Advancements Enhancing Efficiency
Technological innovation is significantly improving the efficiency and reliability of lyophilization processes. Advanced techniques such as controlled ice nucleation, process analytical technology, and quality by design frameworks are enabling better control over freeze drying cycles. These improvements lead to consistent product quality and reduced processing time.
Automation and digital monitoring systems are also transforming operations. Real time tracking of temperature, pressure, and moisture levels helps optimize performance and minimize errors. Artificial intelligence driven analytics further enhance process efficiency by enabling predictive maintenance and cycle optimization.
In addition, the adoption of single use technologies and robotic systems reduces contamination risks and increases operational flexibility. These advancements are helping service providers meet stringent regulatory standards while maintaining cost efficiency.
Segment Insights and Service Trends
The US bulk lyophilization services market is segmented by format, scale, service type, and end user. Bulk tray lyophilization remains the dominant format, accounting for the largest share due to its efficiency in handling large volumes. Shelf freeze drying and hybrid methods are gaining traction for specialized applications that require precision and flexibility.
Commercial and industrial scale operations are expected to witness the fastest growth, reflecting the increasing commercialization of biologics. Full scale commercial bulk lyophilization services hold a significant share, indicating strong demand for comprehensive solutions.
Pharmaceutical and biotechnology companies are the primary end users, driven by their expanding research pipelines and manufacturing needs. Research and academic institutes also contribute to demand, particularly in early stage development and innovation.
Competitive Landscape and Key Players
The competitive environment in the United States is characterized by a mix of established players and specialized service providers. Companies are focusing on expanding capacity, adopting advanced technologies, and forming strategic partnerships to strengthen their market position.
Key players in the US bulk lyophilization services market include:
- Hudson Valley Lyomac
- Alcami Corporation
- Affinity Life Sciences
- OFD Life Sciences
- Quality BioResources
- Symbiosis Pharmaceutical Services Ltd
- Lyophilization Technology, Inc.
- ATTWILL Medical Solutions
- PCI Pharma Services
- Catachem Inc.
These companies are investing in automation, real time monitoring systems, and advanced process capabilities to meet growing demand and maintain competitive advantage.
Emerging Opportunities and Industry Trends
Emerging applications such as cell and gene therapies are creating new growth opportunities for lyophilization services. These therapies require specialized preservation techniques to maintain their effectiveness, leading to increased demand for customized solutions.
Integration with continuous manufacturing and process analytical technology is another important trend. This approach enhances production efficiency, reduces cycle times, and supports large scale manufacturing. Sustainability is also becoming a priority, with companies focusing on energy efficient technologies and environmentally friendly processes.
Additionally, supply chain challenges are encouraging companies to adopt flexible manufacturing strategies and strengthen partnerships with service providers.
Future Outlook
The future of the US bulk lyophilization services market looks promising, supported by strong growth in biologics, vaccines, and advanced therapies. Continuous technological advancements and increasing outsourcing trends will further drive market expansion. The United States is expected to remain a leader in innovation, supported by its well established biopharmaceutical infrastructure and regulatory framework.
Related Reports @
Bupivacaine Liposome Injectable Suspension Market Drivers, Trends, and Forecasts by 2031
Sutherlandia Extract Market Research Report by Size Share Analysis 2031
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Us:
Ankit Mathur | The Insight Partners
E-mail: ankit.mathur@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean| German| Japanese| French| Chinese| Italian| Spanish